Like most physicists, I have a soft spot for Galileo thermometers.

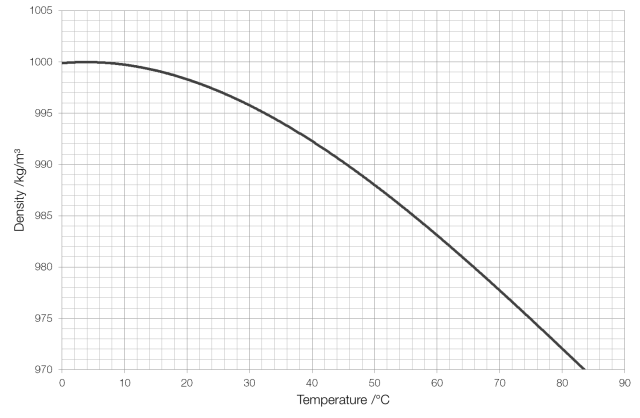
A Galileo thermometer* works because the density of water changes as its temperature changes. The mass of a substance remains constant as it is heated (because the number of atoms doesn’t change) but because those atoms move faster the volume increases and therefore the density decreases, as shown in the graph for water below.
As the water inside the thermometer is heated by its environment its density decreases and the more dense bubbles, that represent lower temperatures, are then more dense than the surrounding fluid and therefore sink. The Galileo thermometer is read by reading the temperature tag of the bubble closest to the middle of the cylinder.
The density of the bubbles inside the thermometer is set by altering the size of the metal tags attached to them: the bubbles that represent higher temperatures have smaller tags and therefore lower densities. (The overall density of the bubble does not change as it is heated because the overall density of the bubble depends only on its overall mass and overall volume, and as the liquid inside a bubble expands it merely compresses the air inside that bubble.)
* Not actually invented by Galileo, but by a group that included one of his students.
I recently moved mine from Michigan to Colorado. It is now only reads 74 degrees – even when i move it to much hotter and cooler environment (outside). Could it be the altitude change? is it fixable? if i get a new one, would you suspect the same problem since i now live at 5800?
Off the top of my head, yes, it could be the altitude. Not much you could do, I’m afraid.
I would suspect that the 78-degree bubble cracked in transit, and it has taken on extra weight. Because there is air-space at the top of the glass tube, if the thermometer is tilted toward horizontal, there can be a ‘flushing’ action where the bubbles are jostled together, and one or more glass bubbles get cracks. I had a 40″ tall Galileo have multiple cracked glass bubbles when it was shipped to me. Fortunately, insurance covered the loss and I received a new one.